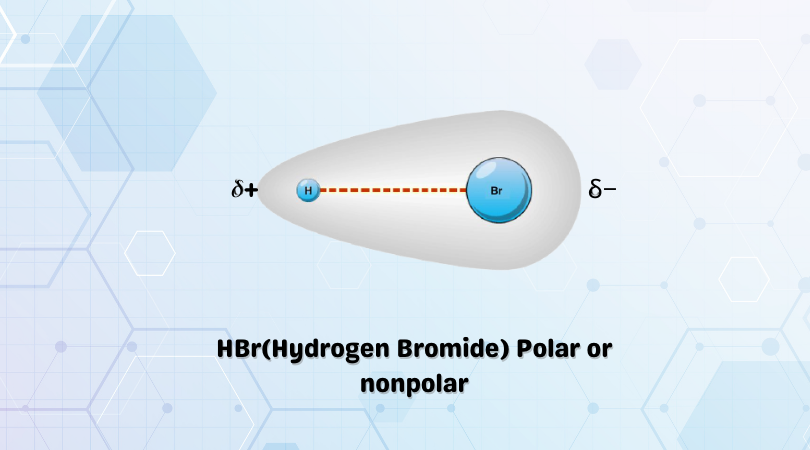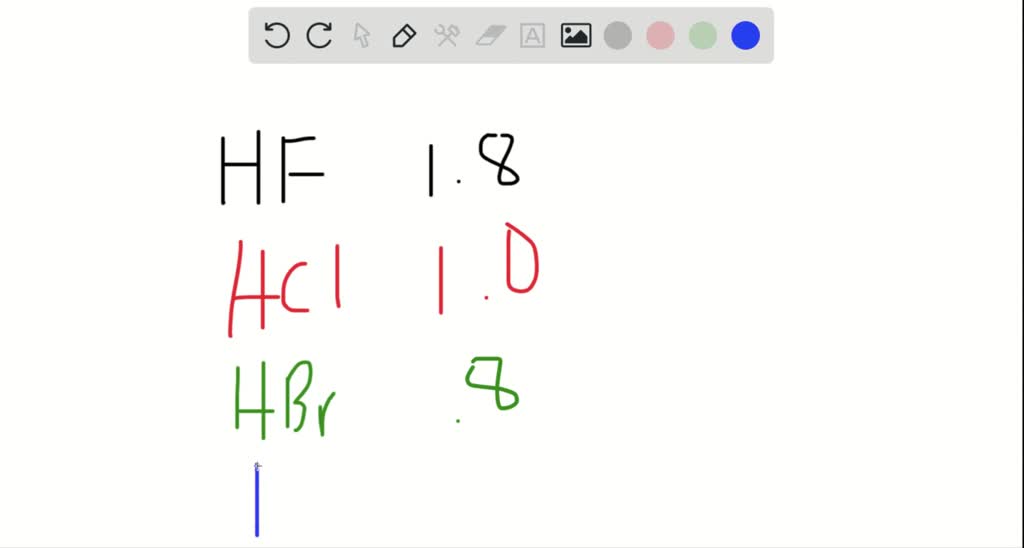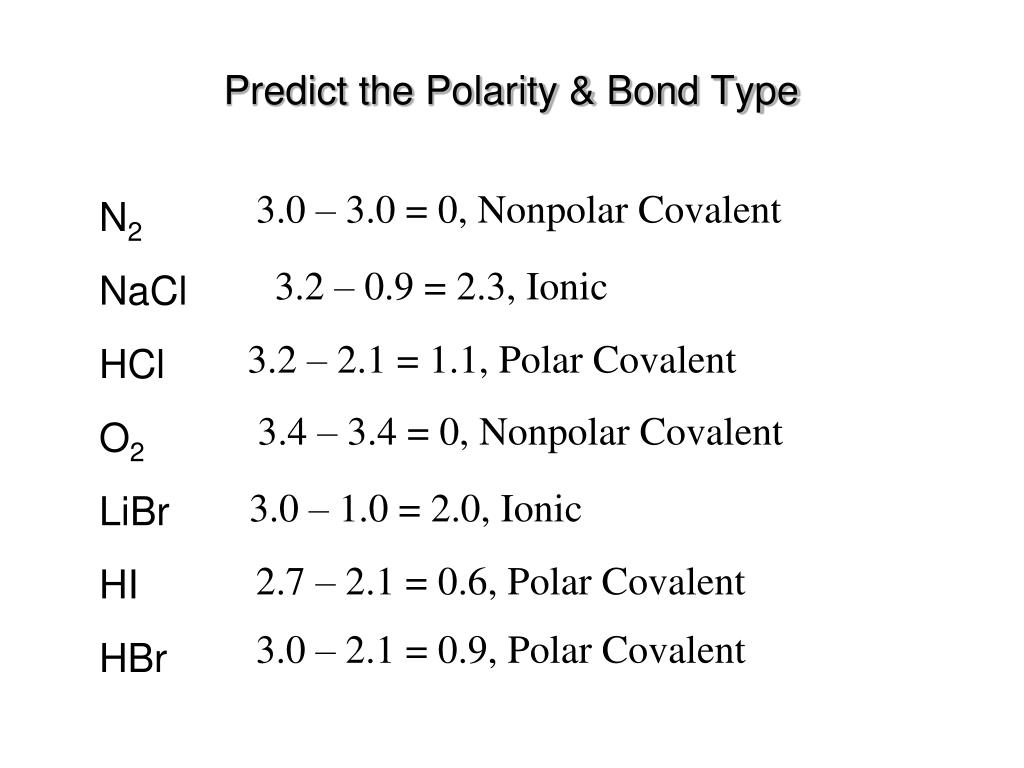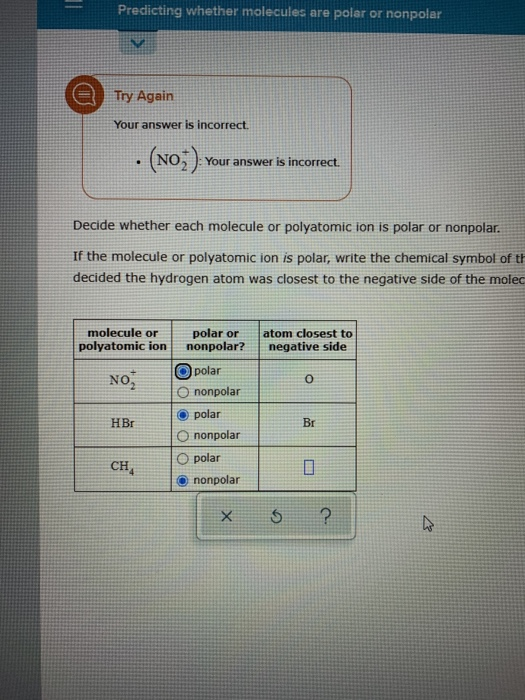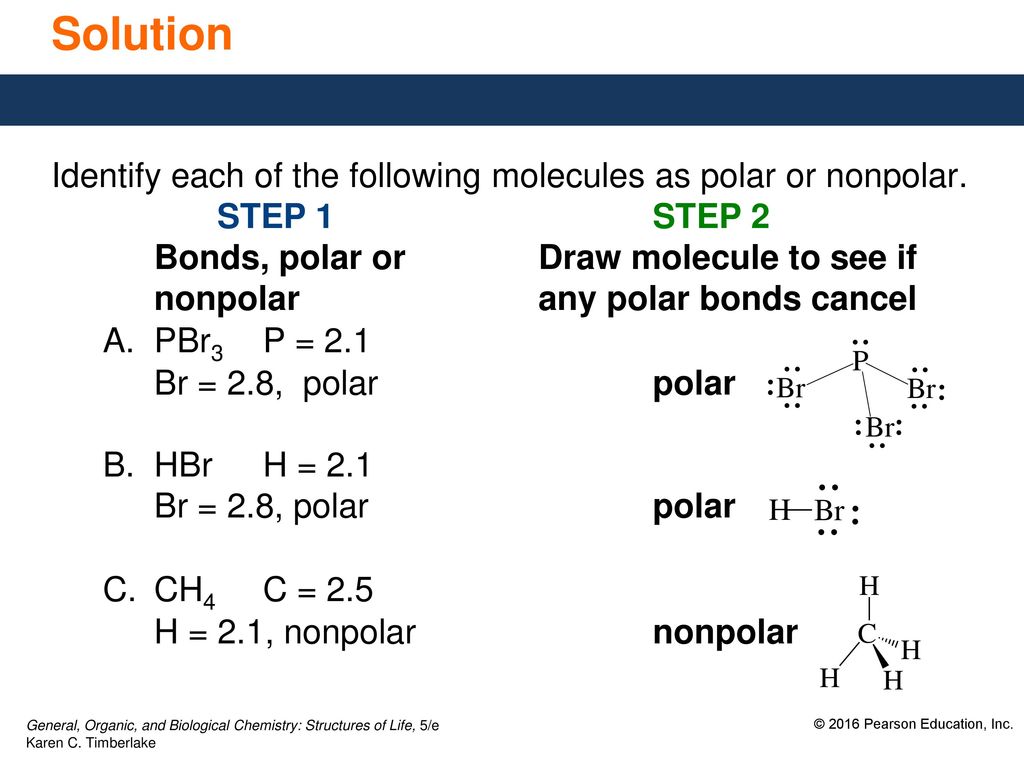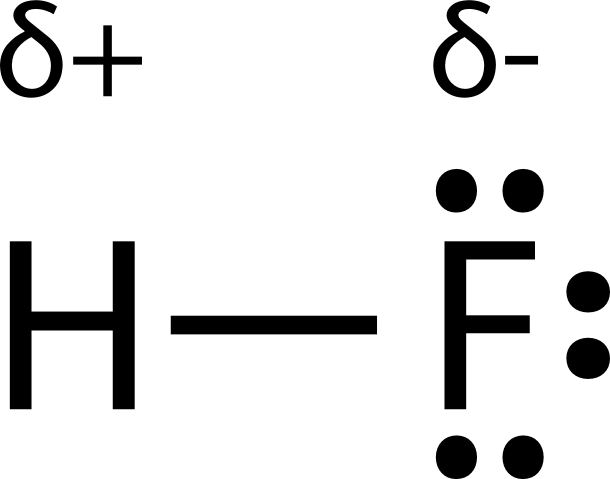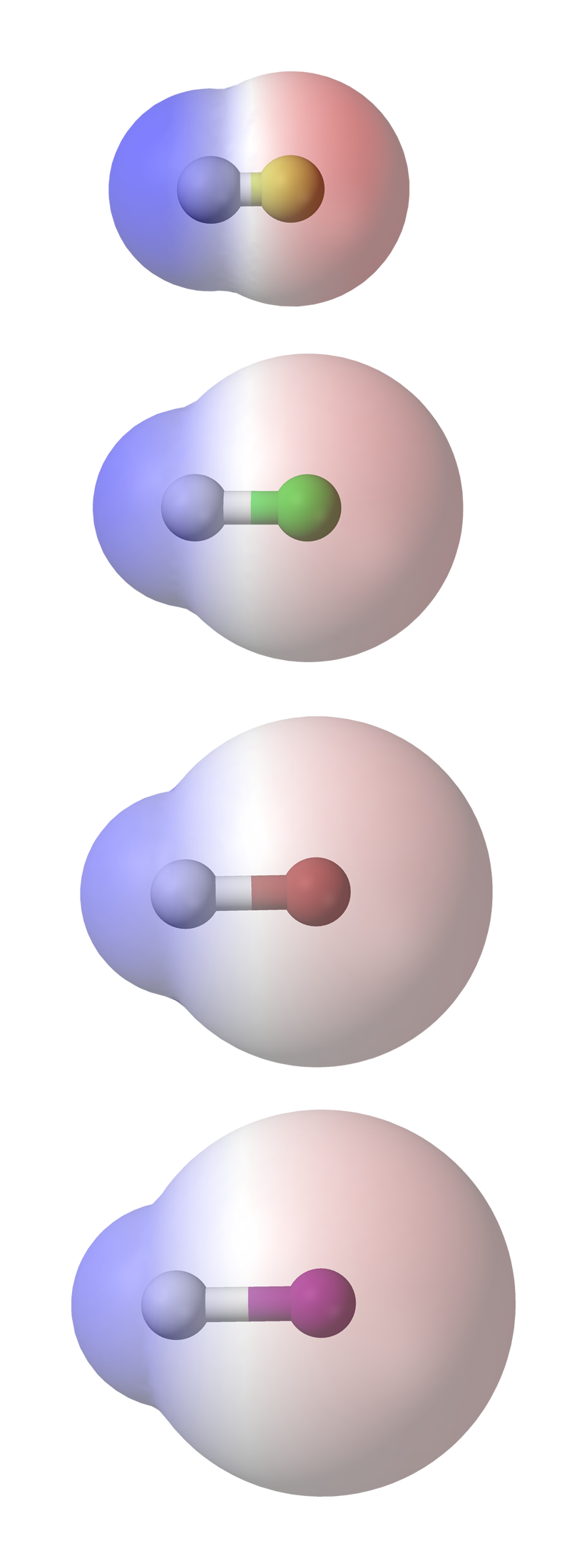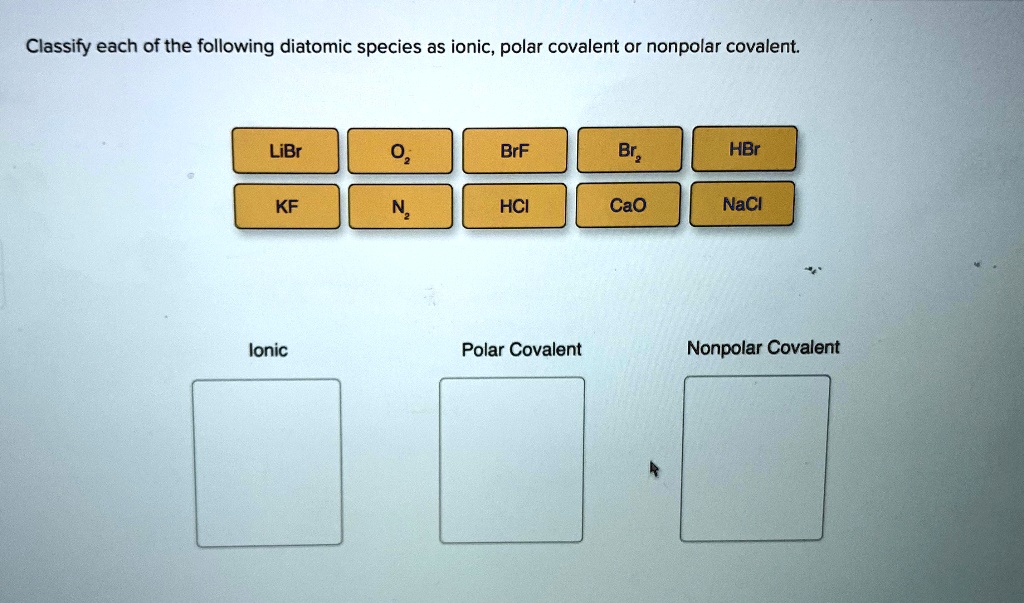
SOLVED:Classify each of the following diatomic species as ionic, polar covalent or nonpolar covalent: LiBr BrF Br; HBr KF HCI Cao NaCl Ionic Polar Covalent Nonpolar Covalent

Bellwork Tuesday Draw the Lewis Structure and name the correct shaped molecule for all of the following. BF3 CH4 HBr NH3. - ppt download